 The element is in the 2nd column of the p block, Group IVA (Column 13).Trending Questions Is octane an ionic compound? What element is too dangerous to add to dilute sulfuric acid? How many valence electrons do the nitrogen family have? What is the oxidation number of Br in BrO? What particle is emitted when P-32 decays to S-32? What are rocks that form when magma cools underground? Is radioactive radiation an element compound homogeneous mixture or heterogeneous mixture? Would the ocular unit increase or decrease as magnification increases? What is this substance name Cl2Ca? How many sulfur atoms are in 0.45 mol BaSO4? Which of the following are elements of the COLDER principle? What is the law of conversation of energy and mass? In the reaction sn plus 2hf - snf2 plus h2 What is the maximum amount of snf2 that can be produced when 0.480 moles of sn are mixed with 0. Germainum is in the 4th row Energy Level of the periodic table. The d orbitals Groups 3-12 (columns) can hold 10 electrons.Įach energy level must be filled before moving up an energy level.Įach orbital group must fill before moving to the next orbital group. What is the most stable monatomic ion formed from caesium What is the group number and number of valence electrons for fluorine What is the electronic configuration for: a. The p orbitals Groups 13 - 18 (columns) can hold 6 electrons Which property would cesium most likely have 1. The s orbitals Groups 1 & 2 (columns) can hold 2 electrons The superscript tells us the number of electrons in the orbital. The Coefficient tells us the Energy Level (Row) of the periodic table You can use this chart to predict whether or not an atom can bond with another atom. The electron configuration for the first 10 elements This is a chart of the most common charges for atoms of the chemical elements. The "f block" on the periodic table are the Lanthanide and Actinide series.Įlectron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table. The "d block" on the periodic table are groups 3-12 make up the d block and the elements' electron configurations end in d. Caesium-137, a fission product, is extracted from waste produced by. The "p block" on the periodic table are groups 13-18 and end in p1, etc. It has only one stable isotope, caesium-133. 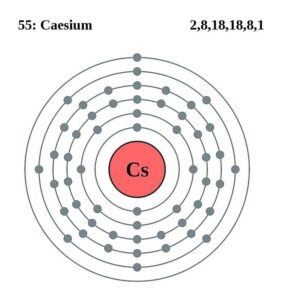
The "s block" on the periodic table are groups 1 and 2 they end in s1 and s2. These rare earth metals are 2 periods behind because the f electrons are even higher in energy than the d electrons. The transition metals are behind by one period because the d electrons are high in energy.įor the rare earth elements (the Lanthanides and Actinides), they end in f. Scandium would end in 3d1, titanium in 3d2, etc. The general rule is that the element's electron configuration ends in d and whatever place they are in. And so it goes.įor the transition metals, groups 3-12, there are many exceptions. In group 4A or 14, all elements end in p2. Group 3A, or 13 all end their electron configurations in p1. 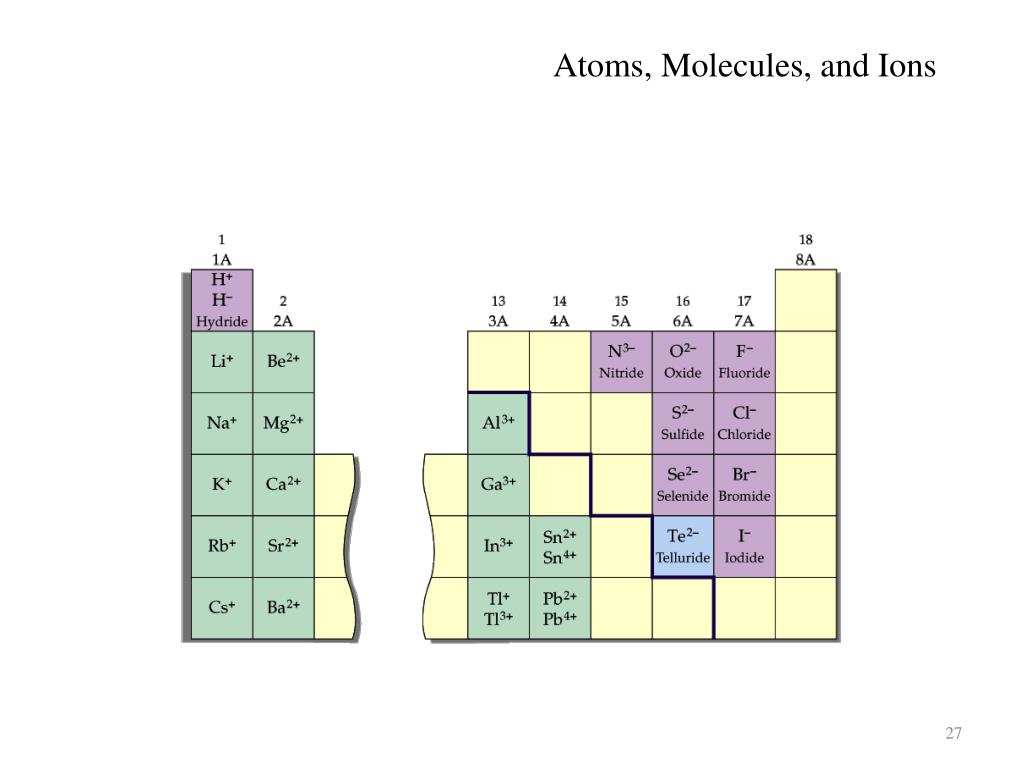
Group 2 elements (2A), the alkaline earth metals, all end in s2 What is the most stable monatomic ion formed from fluorine How many pairs of valence electrons do the bromine atoms have in the following molecules and ions. 
Predict the charge on the most common monatomic ion formed by each element. What period the element is in determines the 1st number.Įxample: H ends in 1s1 (even though H is not a metal, it resides in this group because it also has one valence electron) B The nearest noble gas is krypton, so we predict that selenium will gain two electrons to form the Se 2-ion, which has the same number of electrons as krypton. Group 1A (1), the alkali metals all end is s1. When looking at electron configuration, your fill order of electrons is: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
